- Blog
- Chorando se foi lambada translation
- Train simulator 2014 pc
- Mortal online macro uopilot
- Luxor 2 free game
- Nile cruise aswan to luxor 2 night
- Fsx fspassengers
- Gsplit free download
- Xmlspy 2009 crack
- Iso 14971
- Thalattu ketkuthamma tamil movie
- Dr- butel
- Milkshake font free download google
- Hd movies hollywood download in hindi
- Watch black butler ciel in wonderland part 1
- Ezdrummer drumkit from hell
- Winaso registry optimizer v4-2-0
- Poker wild card five of a kind
- Sandisk ssd u100 24gb firmware
- Serial number for nch wavepad 7
- Visual basic programming
- Who sang lungi dance song
- Wavelab le vs cubase le
- Lyon healy harp price
- Best instagram bot 2020
- Download resetter canon mp198
- Edius 5 tutorial in hindi torrents

To give you an idea of how much this section was overhauled, the requirements have gone from about half a page to nearly a page and a half! This section now dovetails more closely with the ISO 13485:2016 section 8 requirements for feedback, analysis of data and CAPA. Most of the additions to requirements content were made related to production and post-production activities.
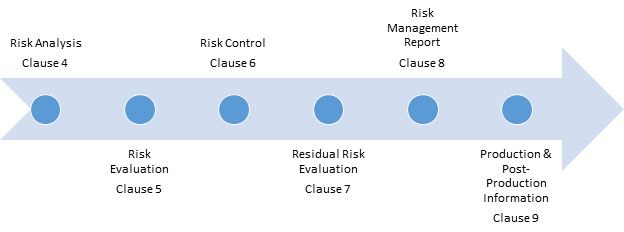
More Requirements for Production and Post-Production Activities That guide defines state of the art as: “Developed stage of technical capability at a given time as regards products, processes and services, based on the relevant consolidated findings of science, technology and experience.” In that context, ISO 14971:2019 could be considered the “state of the art” when it comes to risk management for medical devices. State of the art (3.28) – This perplexing term appears 12 times in the EU MDR and 20 times in the IVDR but is not defined in either regulation! ISO 14971:2019 helps settle the matter by borrowing the newly minted definition found in ISO/IEC Guide 63:2019.The definition now aligns with terminology used in many regulations.

#Iso 14971 professional
This analysis applies to lay users and professional users. “Reasonably foreseeable” can be unintentional or intentional and includes those “why -in-the-world would-someone-do-that” scenarios. Reasonably foreseeable misuse (3.15) – The new definition states that if misuse of a product can result from “predictable human behavior” then you need to take this into account in your risk analysis.Other minor changes were made to the following terms: accompanying documentation, harm, IVD, manufacturer, and use error. Three important definitions have been added to the new ISO 14971:2019 standard and are noted below. Important Definitions Have Been Included in the Current Version of the ISO 14971 Standard The production and post-production section alone is four pages of content. The new ISO/TR 24971:2020 is more than just the new home of the “old” ISO 14971:2007 annexes – it’s been expanded to include more guidance, explanation and examples of the risk management process requirements. You can think of ISO/TR 24971:2020 in the same way you do FDA guidance documents: None of its recommendations are required but you would be foolish not to heed them. (See table below).This was done because changes can be more easily applied to supporting Technical Reports than they can be to standards. One reason for the smaller size, is the old annexes content in ISO 14971:2007 was moved to Technical Report ISO/TR 24971:2020 which itself has been revamped.
#Iso 14971 pdf
If you recently purchased a copy of ISO 14971:2019 you may have noticed that your PDF is much “skinnier.” Sorry to disappoint you but this doesn’t mean the risk management process is simpler. ISO/TR 24971:2020 Is Now an Essential Companion Guide In reviewing the new ISO 14971 standard, pay particular attention to the highlighted sections below. Get up to speed on the changes with our ISO 14971:2019 Risk Management Class.
- Blog
- Chorando se foi lambada translation
- Train simulator 2014 pc
- Mortal online macro uopilot
- Luxor 2 free game
- Nile cruise aswan to luxor 2 night
- Fsx fspassengers
- Gsplit free download
- Xmlspy 2009 crack
- Iso 14971
- Thalattu ketkuthamma tamil movie
- Dr- butel
- Milkshake font free download google
- Hd movies hollywood download in hindi
- Watch black butler ciel in wonderland part 1
- Ezdrummer drumkit from hell
- Winaso registry optimizer v4-2-0
- Poker wild card five of a kind
- Sandisk ssd u100 24gb firmware
- Serial number for nch wavepad 7
- Visual basic programming
- Who sang lungi dance song
- Wavelab le vs cubase le
- Lyon healy harp price
- Best instagram bot 2020
- Download resetter canon mp198
- Edius 5 tutorial in hindi torrents
